
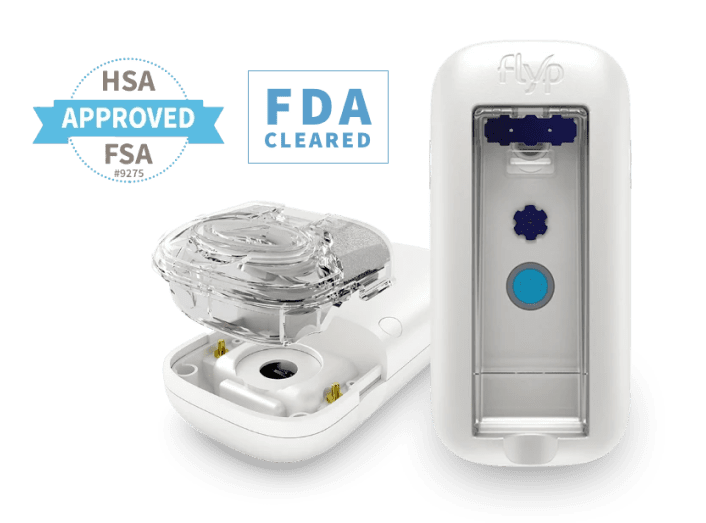
More than 600 million people are affected by asthma and COPD. The nebulizers created by Wellinks help relieve symptoms for those with these conditions.
When Wellinks switched manufacturers, they lost access to important information, which required our team to start the project from scratch. In a very short time frame, we met all customer goals, effectively reproduced the functionality of the previous design while adding innovative new features and wireless technology.
We helped Wellinks reimagine their existing design to produce a portable nebulizer with added features for those who use it.

With no information from the existing manufacturer about the nebulizer, we started this project from scratch on an aggressive deadline.
For user ease, we improved the quality and enhanced the features of the previous design but ensured its appearance and functionality remained the same.
Now, the Wellinks nebulizer boasts wireless capabilities, an electrical hardware redesign, and a firmware redesign. We also developed the back end of the mobile app to integrate with their third-party mobile app team.
While a complete redesign and effective reverse engineering were required, we maintained the original stringent schedule and stayed within the pre-existing FDA certification while making the product even better than before.

Lorem ipsum dolor
Need some help getting your project across the finish line like WellLinks?
Request our FREE design review to ensure your device meets crucial requirements and successfully makes it to market.







Take the guesswork out of the equation with our budgeting calculator and empower yourself to focus on what really matters:
Bringing innovation to life.

This section provides an in-depth look at the redesign process of the Wellinks nebulizer, focusing on the challenges faced during the transition to a new manufacturer. Understanding the complexities involved in such a project is essential for stakeholders interested in medical device innovation.
The redesign not only involved replicating existing functionalities but also integrating advanced features such as wireless technology, which enhances usability and patient experience. This comprehensive overview outlines the critical steps taken to ensure the project met tight deadlines while adhering to regulatory standards.
The Wellinks nebulizer incorporates several innovative features that set it apart from traditional models. These enhancements, including wireless capabilities and improved design elements, significantly improve patient care and ease of use.
By integrating modern technology, such as real-time monitoring and user-friendly interfaces, the Wellinks nebulizer aims to provide a seamless experience for asthma and COPD patients. These innovations not only enhance functionality but also align with the latest trends in medical device development.
The partnership between Wellinks and C3 Medical exemplifies effective collaboration in the medical device industry. This section explores how both teams worked together to overcome obstacles and achieve project goals within a limited timeframe.
Through open communication and a shared vision, the collaboration resulted in a product that not only met but exceeded expectations. This case study serves as a model for future partnerships in medical device consulting, highlighting the importance of teamwork and expertise.
As the medical device industry continues to evolve, the future of nebulizer technology looks promising. This section discusses emerging trends and potential advancements that could further enhance the efficacy and user experience of nebulizers.
With ongoing research and innovation, future nebulizers may include features such as AI-driven medication delivery systems and enhanced connectivity options. Staying ahead of these trends is crucial for companies like Wellinks as they strive to improve patient outcomes and adapt to changing healthcare needs.