If you’re developing a medical device for the first time, one of the earliest and most important steps is defining your user needs. Get this right and the rest of your development process has a solid foundation. Get it wrong and you’ll spend time and money correcting problems that could have been avoided from day one.
This isn’t just a documentation exercise. Medical device user needs are the starting point for nearly every major decision that follows including your engineering requirements, your hazard analysis, your regulatory strategy, and ultimately whether your device actually works the way it’s supposed to in the hands of real people.
Here’s what medical device user needs actually are, how they connect to your design requirements, and why both matter for getting your product to market.
What Are Medical Device User Needs?
Medical device user needs describe your product from the perspective of the people who will actually use it: patients, clinicians, technicians, or caregivers. They capture what your device needs to do in the real world, without locking you into any specific design solution yet.
That last part is important. User needs are written before you’ve committed to how the device will be built. They describe outcomes and behaviors, not components or materials. This keeps your thinking open and user-centered at the stage when it’s still easy to course-correct.
The FDA puts it simply: user needs are about making the right product.
A well-written user needs document covers:
- The intended use of the device
- Proposed indications for use
- The environment in which the device will be used
- A profile of your users
- High-level performance and functionality expectations
- Human factors considerations (for example, whether the device is worn, handheld, or operated remotely)
- Target markets and the regulatory landscape for each
This document also forms the basis for your Hazard Analysis, the process of identifying what could go wrong when someone uses your device. That analysis then feeds directly into your design input requirements.
Who Counts as a “User”?
This is a question early-stage founders often overlook. In medical device development, “users” aren’t just patients. Depending on your device, your user population might include:
Surgeons or interventional specialists Nurses or bedside clinical staff Home care patients or their family members Biomedical technicians responsible for maintenance Hospital administrators involved in procurement or setup
Each of these users may interact with your device differently, in different environments, with different levels of training and physical ability. Your user needs document needs to account for all of them, because your hazard analysis, your human factors studies, and eventually your FDA submission will.
Defining your users narrowly early on is one of the mistakes that tends to surface late in development, when it’s expensive to fix.
How Design Input Requirements Are Different
Once your user needs are defined, design input requirements translate them into specific, measurable engineering criteria. If user needs answer “what does this product need to do for the user,” design input requirements answer “how do we build it to do that.”
The FDA frames it this way: design input requirements are about making the product right.
Think of it as the handoff from the world of the user to the world of the engineer. Everything in your requirements document should be traceable to a specific user need. If you can’t point to a user need that justifies a requirement, that requirement probably shouldn’t be there, or your user needs document is missing something.
Design input requirements can cover a wide range of criteria, including:
Functional and physical specifications Performance and reliability targets Electrical, mechanical, and chemical safety Biocompatibility and toxicology Electromagnetic compatibility Risk management inputs Regulatory and labeling requirements for your target markets Usability and human factors Sterility, packaging, and environmental conditions Manufacturing and installation requirements
Each requirement must be unambiguous and either measurable or verifiable. Tolerances and limits should be defined wherever applicable. No two requirements can conflict with each other, and none can conflict with the industry or regulatory standards that apply to your device.
These documents are refined and updated as development progresses. They’re living records of your design decisions, and they’re exactly what regulators will scrutinize during a submission review.
How User Needs and Design Input Requirements Work Together
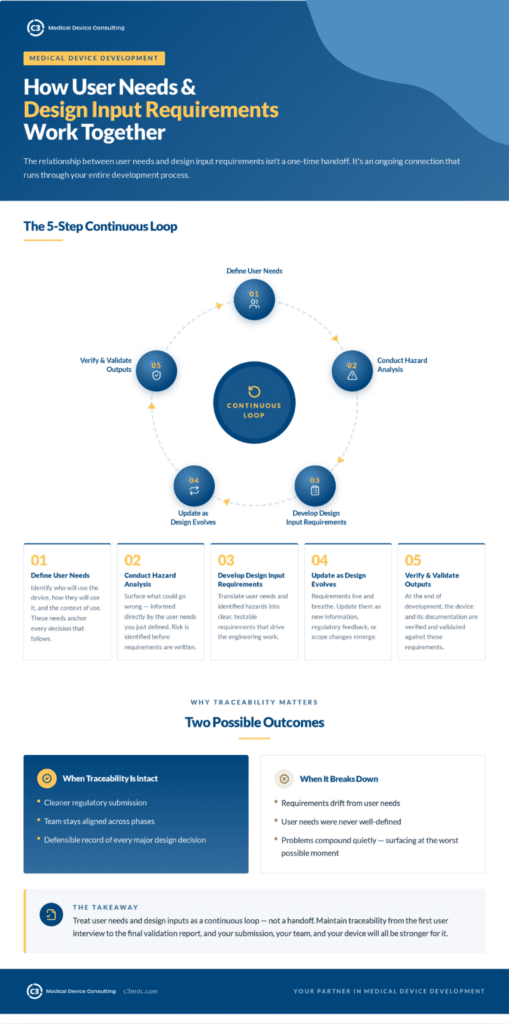
The relationship between user needs and design input requirements isn’t a one-time handoff. It’s an ongoing connection that runs through your entire development process.
Here’s a simplified version of how it works in practice:
You define your medical device user needs based on who will use the device, how, and in what context. You conduct a Hazard Analysis to identify what could go wrong, informed directly by those user needs. You develop design input requirements that address both the user needs and the hazards identified. As your design evolves, your requirements are updated to reflect new information, regulatory feedback, or changes in scope. At the end of development, your design outputs, the actual device and its documentation, are verified and validated against those requirements.
When this traceability is intact, your regulatory submission is cleaner, your team stays aligned, and you have a defensible record of every major design decision. When it breaks down, when requirements drift from user needs, or user needs were never well-defined to begin with, the problems tend to compound quietly until they surface at the worst possible moment.
How User Needs & Design Input Requirements Work Together
Why This Matters for Early-Stage Founders
Skipping or rushing through user needs is one of the most common and costly mistakes in early medical device development. Without a clear picture of who is using your device and how, your engineering decisions have no anchor. Scope creeps. Requirements shift. And by the time you’re preparing for a regulatory submission, you’re filling in gaps that should have been addressed at the very beginning.
The pressure to move fast is real. Funding timelines, competitive windows, and investor expectations all push founders toward speed. But cutting corners on user needs doesn’t actually save time, it borrows it from later in the project, at a much higher interest rate.
A thorough user needs document from the start keeps your team aligned, your development on track, your regulatory pathway cleaner, and your costs more predictable. It’s one of the highest-leverage investments you can make in the early stages of a device program.
It also sets the tone for how you work. Founders who take user needs seriously tend to make better design decisions throughout development, because they’ve already done the hard thinking about who they’re building for and why.
Common Mistakes to Avoid
A few patterns we see regularly with early-stage teams:
Writing requirements instead of needs.User needs should describe what the device must accomplish for the user, not how it will be built. If your user needs document reads like an engineering spec, it’s probably too deep too early.
Defining users too narrowly.As covered above, your user population is often broader than it first appears. Spend time on this before you finalize the document.
Treating it as a one-time task.User needs and design input requirements are living documents. They should evolve as you learn more — through testing, regulatory feedback, and clinical input.
Skipping the hazard analysis connection.The Hazard Analysis is the bridge between user needs and design inputs. Treating these as separate workstreams instead of a connected process creates traceability gaps that are difficult to close later.
Going it alone.If your team doesn’t have deep experience with FDA design controls, working with engineers who do can save you from mistakes that are expensive to unwind mid-development.
Ready to Define Your Medical Device User Needs?
If you’re at the early stages of development and want a partner who will work through this process with you — not hand you a template and walk away —book a conversation with our team. We’ve been doing this work directly with founders and engineering leaders for more than 16 years, exclusively in medical devices.
You can also use ourdevelopment cost estimatorto get a clearer sense of what a structured development program might look like for your device.














